 
Idarucizumab is a humanized monoclonal antidabigatran antibody fragment approved for reversing dabigatran-associated bleeding. Two FDA-approved target-specific reversal agents are now commercially available. These agents have a number of approved indications, including prevention of systemic embolization and stroke in patients with nonvalvular atrial fibrillation, preventing and treating venous thromboembolism, and secondary prevention of arterial ischemic conditions in chronic coronary arterial disease and peripheral artery disease ( Table 1). 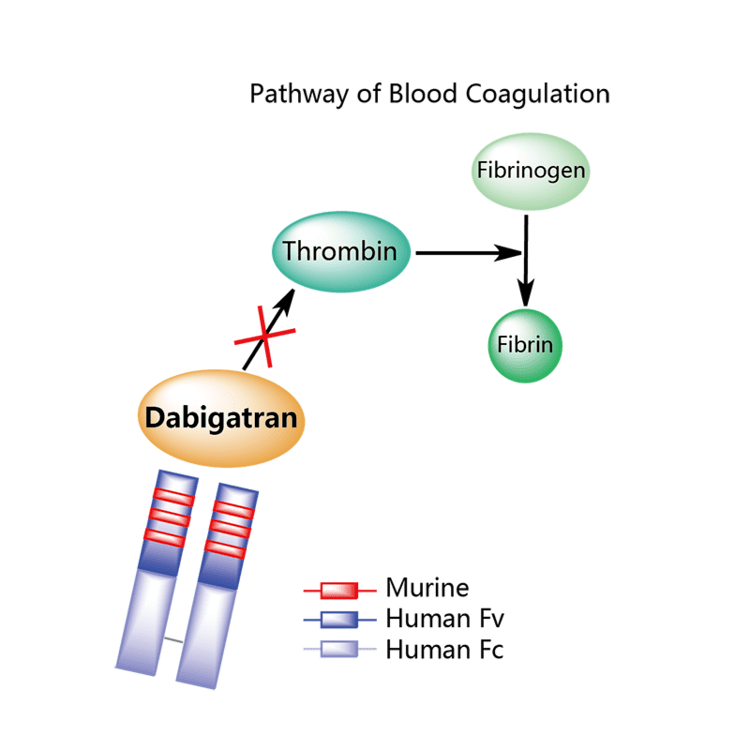
These agents are expensive and pose some thrombotic risk-thus the need for comprehensive reversal guidelines.ĭirect oral anticoagulants (DOACs) include dabigatran, which is a direct thrombin (factor IIa) inhibitor, and 4 direct factor Xa inhibitors: rivaroxaban, apixaban, edoxaban, and betrixaban. Reversal agents are now commercially available and have demonstrated their ability to reverse the effects of DOACs. The number of patients treated with DOACs is increasing, as are rates of major and life-threatening DOAC-associated bleeding.Ĭlear guidelines for the reversal of DOAC-associated bleeding are needed. according to expert consensus, the combination of idarucizumab and IVT is suggested f or patients with acute ischemic stroke of administration of thrombolysis after prior neutralization of the effect of dabigatran with the antidote PRAXBIND is possible (indications for Praxbind include emergency procedures that may involve thrombolysis). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
